Our research interests are autoimmunity, host-microbe interactions, neonatal immunity, and nutritional immunology. There are 3 main directions of research in our laboratory. The first is to determine the role of gut microbiota in the pathogenesis of lupus. We have found that a leaky gut drives autoimmunity, and we are currently delineating the underlying mechanisms. The second direction is to reveal novel mechanisms of microbiota-mediated regulation of neonatal immune development, and how immunological imprints during the neonatal stage would impact the development of autoimmunity later in life. The third direction is focused on the role of vitamin A in the pathogenesis of lupus.
Gut Microbiota and Autoimmunity
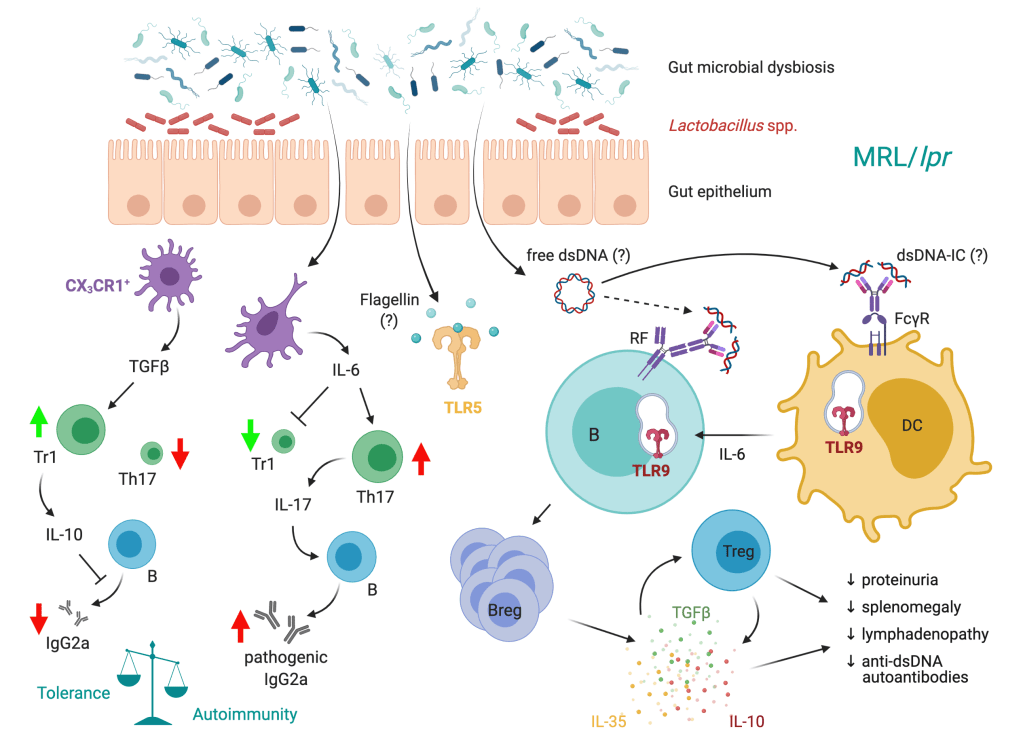
Systemic lupus erythematosus (SLE) is a complex autoimmune disorder with no known cure. Increasing evidence in recent years suggest that microbiota—largely commensal bacteria living in our gut—and the immune system interact to maintain tissue homeostasis, but the involvement of this interaction in the pathogenesis of SLE remains unclear. Our research team was the first to describe changes of gut microbiota in lupus mice vs. healthy controls, with lupus mice having a decrease of Lactobacillaceae and an increase of Lachnospiraceae. Based on this observation, we administered a mixture of 5 strains of Lactobacillus spp. into lupus mice via oral gavage, which significantly attenuated the disease. This follow-up study, which was done using MRL/lpr mice, along with two recent studies using additional models of murine lupus, established gut microbiota as a cause, instead of effect, of SLE pathogenesis. Our ultimate goal is to mechanistically define the role of gut microbiota in the pathogenesis of SLE.
Microbiota at Maternal-Neonatal Interface
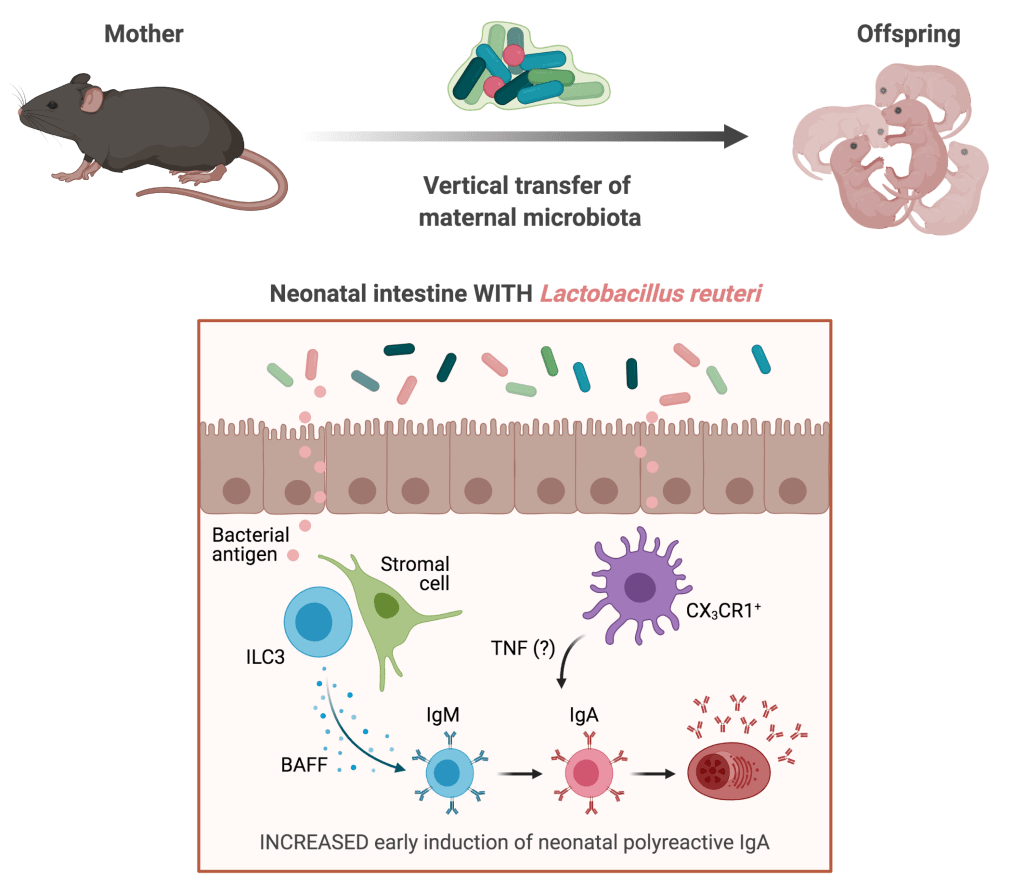
Infants are prone to enteric infections due to an underdeveloped immune system. Maternal microbiota, through shaping the neonatal microbiota, helps establish a strong immune system in infants. We and others have observed the phenomenon of enhanced early neonatal IgA production in pre-weaning immunocompetent mice nursed on immunodeficient dams. Here, we show that this enhancement of IgA in neonates results from maternally derived microbiota. In addition, we have found that the neonatal IgA production can be induced by Lactobacillus reuteri, which is enriched in the milk of immunodeficient dams. Moreover, we show that while the production of neonatal IgA is dependent on neonatal T cells, the immunodeficient maternal microbiota-mediated enhancement of neonatal IgA has a T cell-independent component. Indeed, this enhancement may be dependent on type 3 innate lymphoid cells in the neonatal small intestinal lamina propria. Interestingly, maternal microbiota-induced neonatal IgA does not cross-react with common enteric pathogens. Future investigations will determine the functional consequences of having this extra IgA.
Vitamin A and Autoimmunity

Vitamin A plays important roles in a wide range of biological processes. Among these is immune modulation, where vitamin A is required for the development and maintenance of a functional immune system. Most effects of vitamin A are exerted by its active metabolite, retinoic acid (RA), which through interaction with its receptors (RAR) controls the transcription of RA-responsive genes. In the past 10 years, much interest has been focused on the role of RA on the intestinal mucosa. The current literature emphasize that RA maintains gut homeostasis by regulating gut-homing of many immune cell types. However, the effect of RA on intestinal epithelial cells (IECs) has been less well studied. The function of IECs as regulators of gut barrier function mandates a more complete analysis of these cells. Leaky gut is present in numerous autoimmune diseases, and may exacerbate the autoimmune state. RA may reverse a leaky gut either (or both) by: 1) direct regulation of the intestinal epithelium, and/or 2) modulation of gut microbiota. We hypothesize that RA affects multiple types of IECs and enhances intestinal epithelial homeostasis both directly and through increasing butyrate-producing commensal bacteria in the gut.
Funding Acknowledgements


